Behavioral Science Essays
The
Attentional Blink
The
Glutamate Theory of Schizophrenia
Non-intelligent
Perception
Anxiety
and the GABA-A receptor subunit
On
The Theory-Ladenness of Observation
ECT in Depressed
Older Adults
Dennett Indented?
On the Theory-Ladenness of Perception, Connectionist Models, Plasticity, and the Reentrant Neural Pathways of the Brain
Sten M. Andersen
Whether or not observation is theory-laden has important bearings upon the way in which we view the scientific process. If all observation is theory laden, we might have to conclude that we can never discover truth; that the results of all scientific endeavours are mere isomorphic representations of some postulated "real world," i.e., relativism. In this essay, I will address the question of whether observation is necessarily theory-laden, by investigating the issue with respect to current neurobiological understanding of the brain. Specifically, I will look at two arguments (both due to Churchland) for the theory-ladenness of perception, the first coming from connectionist modelling, thus being a theoretical approach, the other drawing upon findings from biological neuroscience: that the brain has massive reentrant connections going from the higher to the lower levels, all the way out to, for example, the retina. An attack on the connectionist argument will be encountered, but shown to be wrong. An attack on the second, biological argument will also be encountered, arguing that the role of the massive reentrant connections in the brain is not to impose a theory on the input, but is rather to make the peripheral systems part of higher cognitive functions. Against this view, it will be argued that the two positions are not mutually exclusive. Finally, the plasticity of the brain will be looked at. It will be concluded that perception is theory-laden.
It is widely held that observation is theory-laden. This view is clearly underlying a statement such as "each of us literally chooses, by his ways of attending to things, what sort of universe he shall appear himself to inhabit" (James, 1890). A strong case has been made that many aspects of the scientific process are influenced by theory, e.g., attention, data interpretation, data production, and scientific communication (Brewer & Lambert, 2001). To add to this list, Churchland (1989) argued for a strong form of theory-ladenness in perception. I now turn to explaining the theoretical argument for the cognitive penetrability of perception, i.e., I will try to explain why, according to Churchland, perception is theory-laden. The argument involves a somewhat detailed understanding of the connectionist model.
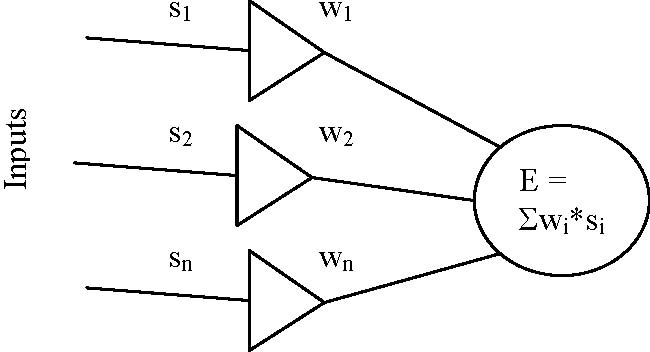
The connectionists model the brain as populations of artificial neurons. These artificial neurons are, in essence, mathematical statements that are supposed to capture the essential workings of proper neurons. An artificial neuron (i.e., a mathematical function), receives as input what is output from other artificial neurons, weighted by the strength of the connection between the two neurons. That is, the total input E to a neuron is the sum of all inputs weighted by the strength of the respective synapses, E = Σwi*si, where si is the strength of the input and wi is the weight of the synapse (McCulloch & Pitts, 1943; Rosenblatt, 1958; Rumelhart & al., 1986), see Fig.
1.
Transfer s0 function
Output
Figure 1. A model of a neuron The output of such a neuron is usually not "all-or-nothing", as is often said of real neruons. Rather, the weighted sum E is passed through a transfer function, usually a sigmoidal (that is, S-shaped) function that "squashes" the output value. That is, if we use a sigmoidal function (like the logistic function), we approximate ”a threshold behaviour while providing a continous output function” (Luger & Stubblefield, 1998,
p. 672). Output from the neuron then becomes s0 = f(Σxiwi) = 1 / (1 + e-λ* Σxiwi) (Rumelhart et al., 1986). A neuron may be at the input layer, which receives data from the outside world; it maybe at the output layer, sending information to the world; or it may be in between the input and output layers, in a “hidden” layer. Usually, every neuron in layer L is connected to every neuron in both layer L-1 and in layer L+1 (see Fig. 2). In a backpropagation network, the error of the output is calculated and backpropagated through the network during training. That is, the difference between the network's output and the expected output is calculated, and the weights that were most responsible for the error are modified accordingly. Hence, the system learns by adjusting the weights. Now, the important thing here is what such networks are capable of learning during training. It has been shown that networks of this type can learn to discriminate between environmental features that humans also discriminate between, but which are difficult to describe by their physical properties (Churchland, 1989) (for examples, see Gorman & Sejnowski, 1988 and Rosenberg & Sejnowski, 1987). They thus seem to have something very special in common with biological brains.
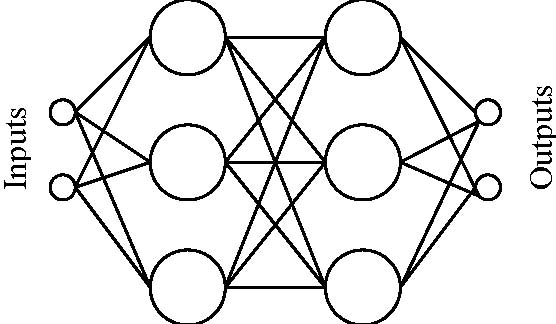
Figure 2. A two hidden-layer network
Even though the network described above shares some characteristics of biological brains, it is in many ways not biologically realistic, and (Churchland, 1989) acknowledges this. For example, in the artificial neural network (ANN), all neurons in one layer connect to all neurons in the next layer, whereas in real brains, an axon synapses with only a fraction of the cells in the target population. Also, in real brains, neurons seem to be either excitatory or inhibitory; in the ANN, it is crucial that a neuron can excite some target neurons while at the same time inhibiting others for the network to work. Nevertheless, such artificial networks function as proof-of-concept: networks can be made that performs similar tasks to the ones humans perform in seemingly similar ways. Thus, if the neural networks of biological brains are neural networks in the sense of the connectionist, then the way biological brains learn is by updating the weight between neurons. And this is the crux of Churchland's (1989) claim that perception is theory-laden: the signals that reach the brain from the sensory transducers will necessarily be propagated from neuron to neuron, and the weights between these neurons are the result of prior experience; hence, prior experience influences what is perceived.
If Churchland (1989) is right that prior experiences influence perception in the same way as it does cognition, we would expect perception to "learn" from its mistakes; perception should, to some extent, be "smart." That perception actually is smart has been argued by several researchers (e.g., Gregory, 2001; Kennedy, 2001; Rock, 2001), but all these researchers fail to show any signs of learning in perception. Quite contrary, on their accounts, perception seems to resist learning, as becomes evident when one considers, for example, visual illusions. Even after having measured the lines in a Muller-Lyer figure and having found them to be of the same length, they are still perceived to be different. Visual illusions persist, even for a lifetime (Gregory, 2001). This is not what would be expected from an ANN (Fodor, 1984). If the weights between neurons were updated all the time, we would surely expect them to learn the true properties of these lines, and see that they are identical in length?
Maybe learning the properties of the world that lead to such illusions as the Muller-Lyer is exactly what neurons in the early parts of visual processing do. Instead of being the result of rigidity, such visual illusions might be due to having learned to make corrections in judging absolute size (Gregory, 1978). If this is correct, then merely measuring the lines of the Muller-Lyer illusion every now and then will not be enough to counter the everyday meetings with objects in the real world that make the illusion useful. But we need not content ourselves with hypothetical entities. A more dramatic effect of perceptual learning has been shown by several researchers (summarised by Kottenhoff, 1957). After participants had worn spectacles turning the world upside-down for a week, the world was not perceived to be upside-down anymore. When the spectacles were subsequently removed, participants were disoriented just like they had been when the spectacles were first put on. Thus, it does seem like perceptions are altered with experience. In other words: prior experience influences what is perceived, consistent with the connectionist model. Thus, it has been shown that the theoretical model can predict properties of real brains. I now turn to findings from neuroscience that also seems to indicate that perception is theory-laden.
Cell-staining techniques have shown that the main visual pathways start in the retina, go through the optic nerve to the lateral geniculate nucleus, the primary visual cortex, the secondary visual cortex, and from there to several higher areas; these pathways are called ascending, as signals are seen as travelling up a hierarchy of increasingly complex processing-units (Merigan & Maunsell, 1993). Similarly, there are descending pathways, seemingly going all the way back to the retina in the visual streams (Van Essen, 1985; Zeki, 1978), and likewise for the other modalities (Livingstone, 1978). It would be natural to believe that the function of these descending pathways is to allow "centrifugal control" (Churchland, 1989, p. 266), letting higher cognitive functions modulate lower level activity, thus, being another route for theories to adjust what is perceived.
The view that the role of the reentrant pathways (i.e., the descending pathways) is to allow control of perception from higher levels has been contented by Raftopoulos (2001), who argues that "the role of the descending pathways is to enable the early-vision processing modules to participate in higher-level cognitive functions" (p. 188). It is motivated by the findings that whereas only 18% of the neurons in the primary cortex change their responses according to the image perceived, 50% do this halfway up the (ventral) visual stream, and almost 100 % in the inferotemporal (IT) cortex (Leopold & Logothetis, 1996), which is regarded as the end of the ventral processing stream. He takes this to indicate that the early neurons are little more than messengers, conveying what ever comes from the retinas. If this interpretation is true, then what are the reentrant pathways for? In short, Raftopoulos (2001) argues that since visual imagery activates the same brain areas as perception (Damasio, et al., 1993; Farah, 1984), and that upon recognition of an object, these areas are activated with delay; that is, the same areas as are activated upon passive perception of something, are activated again about 250 ms later, this time being part of the semantic processing. Thus, the task of the reentrant pathways is to amplify processes already going on in the lower levels of the stream, making them part of the higher-level cognitive functions. It is outside the scope of this essay to explicate his argument; the interested reader is referred to the original article. However, some problems with the underlying assumptions will be pointed out.
As we have seen, Raftopoulos (2001) used the findings of Leopold and Logothetis (1996) (that an increasing number of neurons respond to the image perceived as we move up the processing hierarchy,) to argue that early neurons are little more than messengers, conveying what ever comes from the retinas. This interpretation should not be taken at face value. Polonsky, Blake, Braun, and Heeger (2000) used fMRI to investigate binocular rivalry. If two incompatible monocular images are presented to the two eyes, rivalry will occur; only one of the images will be perceived at one point in time. Polonsky et al. found that rivalry-related signal fluctuations in the primary visual cortex (V1) were about equal to those observed in higher visual areas (V2, V3, V4), thus indicating that binocular rivalry might start as early as V1. If this is correct, early neurons are not mere passive messengers, but are actively engaged in the processing of visual information. It is also informative to look at Leopold and Logothetis's (1996) own interpretation of their data. They believe that the activity changes that are actually found in early visual cortex are reflections of the monkey's percept (hence the title of their paper). Also, von der Heydt, Peterhans, and Baumgartner (1984) found that some V1 cells respond to illusory contours. Let me say it again: early neurons are not mere messengers; they perform processing.
However, it is quite congruent to believe that the reentrant pathways impose some theory on perceptions, that early neurons learn and are theory-laden in that way also, and that the early areas are part of higher level processes. For example, Tong and Engel (2001) argue that V1 is "important in the selection and expression of conscious visual information" (p. 195), and that the result of binocular rivalry is feedback inhibition from V1 to the lateral geniculate nucleus (LGN) (i.e., information-flow in a descending pathway). If you think about it, it would actually be quite puzzling if the brain did not rewire when meeting new information, given its plasticity. When two fingers on a monkey were sewn together, the cortex representation fused to form a single superfinger (Merzenich, 1988); when trained to trail a finger on the edge of a spinning disc, the mapping of the finger grew, stealing cells from neighbouring areas mapping the monkey's face. Burning a small hole in the back of a cat's eyeball with laser resulted in the cortex redrawing its boundaries within minutes (Gilbert & Wiesel, 1992). Rewiring the input from the retina of a newborn ferret through the MGN (medial geniculate nucleus) instead of the LGN, thus leading visual information to the auditory cortex, lead to a development of the auditory cortex similar to what would be expected from a normal visual cortex: cells with directional selectivity, orientation selectivity, and edge-detecting capabilities (Sur, Garraghty & Poe, 1988).
Thus, it would be very strange indeed if the cells of the early visual areas were not equally plastic. It seems like the burden of proof have been shifted to those who propose that they are not.
We have seen that the connectionist model leads to predictions of high plasticity throughout the brain, and that a strong form a theory-ladenness in perception follows if this model is correct. An attack on the model involving one type of visual illusions has been encountered, but was found wanting, as these visual illusions might be explained by a learning process, exactly what they were meant to be counterexamples of. Evidence from neuroscience involving reentrant pathways was also looked at; it was voiced that these pathways might be involved in centrifugal control of the peripheral systems, i.e., a weaker sort of theory-ladenness. An attack on this view was countered. Finally, it was argued that the plasticity found throughout the brain is a good indicator of it being an ever-learning entity, and thus at every one-time full of anticipations of the world-to-be-perceived. To put it in other words: if the brain had no theories about the world, or, if the brain had preconceived, hardwired theories about the world, then there would be no need for it to change according to new information, as it would not matter anyway (except for storing episodic memories). Thus, since the brain does rewire itself according to new information, we can conclude that this rewiring is a part of updating the brain's hypothesis about the world, hypothesis that will be used in interpretations of new sensory input, that again will lead to updating of hypothesis, in a life-long circle.
References
Brewer, W. F., & Lambert, B. L. (2001, Sept). The Theory-Ladenness of Observation and the Theory-Ladenness of the Rest of the Scientific Process. Philosophy of Science, 68(3), 176.
Churchland, P. M. (1989). A neurocomputational perpsective: The nature of mind and the structure of science. Cambridge, Massachusetts: The MIT Press.
Damasio, H., Grabowski, T., Damasio, A., Tranel, D., Boles-Ponto, L., Watkins, G. L., et al. (1993). Visual recall with eyes closed and covered activates early visual cortices. Society for Neuroscience Abstracts, 19, 1603.
Farah, M. J. (1984). The neurological basis of mental imagery: A component analysis. In S. Pinker (Ed.), Visual Cognition (pp. 245-271). Cambridge, MA: The MIT Press.
Fodor, J. A. (1984). Observation reconsidered. Philosophy of Science, 51(1), 23-43.
Gilbert, C. D., & Wiesel, T. N. (1992). Receptive-field dynamics in adult primary-visual cortex. Nature, 356, 150-152.
Gorman, R. P., & Sejnowski, T. J. (1988). Learned classification of sonar targets using a massively-parallel network. IEEE Transactions: Acoustics, Speech, and Signal Processing.
Gregory, R. L. (1978). Space in pictures. In C. F. Nodin & D. F. Fisher (Eds.), Perception and Representation (pp. 228-245). New York: Praeger Publishers.
Gregory, R. L. (2001). Action potentials to potential actions. In T. E. Parks (Ed.), Looking at looking: An introduction to the intelligence of vision (pp. 51-71). Thousand Oaks: Sage Publications.
James, W. (1890). Principles of Psychology. Vol. 1. New York: Henry Hold and Company.
Kennedy, J. M. (2001). Smart geometry! In T. E. Parks (Ed.), Looking at looking: An introduction to the intelligence of vision (pp. 31-49). Thousand Oaks: Sage Publications.
Kottenhoff, H. (1957). Situational and personal influences on space perception with experimental spectacles. Acta Psychologica, 13, 79-97.
Leopold, D. A., & Logothetis, N. K. (1996). Activity changes in early visual cortex reflect monkeys' percepts during binocular rivalry. Nature, 379, 549.
Livingstone, R. B. (1978). Sensory Processing, Perception, and Behaviour. New York: Raven Press.
Luger, G. F., & Stubblefield, W. A. (1998). Artificial Intelligence. Structures and strategies for complex problem solving (3rd ed.).. Sydney: Addison Wesley Longman, Inc.
McCulloch, W. S., & Pitts, W. (1943). A logical calculus of the ideas immanent in nervous activity. Bulletin of Mathematical Biophysics, 5, 115-133.
Merigan, W. H., & Maunsell, J. H. (1993). How parallel are the primate visual pathways. Annual Review of Neuroscience, 16, 369-402.
Merzenich, M. M. (1988). Representational plasticity in somatosensory and motor cortical fields. Biomedical Research, 10, 85-86.
Polonsky, A., Blake, R., Braun, J., & Heeger, D. (2000). Neuronal activity in human primary visual cortex correlates with perception during binocular rivalry. Nature Neuroscience, 3(11), 1153-1159.
Raftopoulos, A. (2001). Reentrant neural pathways and the theory-ladenness of perception. Philosophy of Science, 68(3), 187-199.
Rock, I. (2001). Stupid perceptions? In T. E. Parks (Ed.), Looking at looking: An introduction to the intelligence of vision (pp. 3-29). Thousand Oaks: Sage Publications.
Rosenberg, C. R., & Sejnowski, T. J. (1987). Parallel networks that learn to pronounce English text. Complex Systems, 1, 145-168.
Rosenblatt, F. (1958). A probabilistic model for information storage and organization of the brain. Psychological Review, 65, 386-408.
Rumelhart, D. E., McClelland, J. L., & The PDP Research Group. (1986). Parallel Distributed Processing. Vol. 1.. Cambridge, MA: The MIT Press.
Sur, M., Garraghty, P., & Poe, A. (1988). Experimentally induced visual projections into auditory thalamus and cortex. Science, 242(4884), 1437-1441.
Van Essen, D. C. (1985). Functional organization of primate visual cortex. In A. Peters & E. G. Jones (Eds.), Cerebral Cortex, Vol. 3. (pp. 259-329). New York: Plenum.
von der Heydt, R., Peterhans, E., & Baumgartner, G. (1984). Illusory contours and cortical neuron responses. Science, 224, 1260-1262.
Zeki, S. M. (1978). Uniformity and diversity of structure and function on rhesus monkey prestriate visual cortex. Journal of Physiology, 277, 273-290.